
The Lantern Project from Sanofi Genzyme and PerkinElmer expands access to free diagnostic genetic testing for a number of lysosomal storage disorders where tens of thousands go undiagnosed
Patients with rare genetic diseases can face a difficult path to a proper diagnosis. Because there is a lower awareness of these diseases and they have symptoms which may initially be confused with more common diseases, diagnosis can take several years with patients often consulting with multiple physicians. Access to comprehensive diagnostic testing can also be a barrier to diagnosis.
The Lantern Project aims to change that by providing a no-cost testing program for patients in the United States who suffer from specific types of lysosomal storage disorders (LSDs). Physicians will be able to use the Lantern Project to arrange for screening of certain suspected LSDs as well as confirmatory DNA testing and phlebotomy services for their patients.
Launched by Sanofi Genzyme with PerkinElmer Genomics, The Lantern Project will assist patients whom physicians suspect may be suffering from Gaucher disease, Fabry disease, Pompe disease, mucopolysaccharidosis type I (MPS I), or acid sphingomyelinase deficiency (ASMD), also known as Niemann-Pick disease types A and B. Additionally, there is the option of an enzyme panel for seven mucopolysaccharidoses and a 105 gene panel for limb-girdle muscular dystrophies (LGMD) and other myopathies.
“The Lantern Project is another reflection of Sanofi Genzyme’s continuing commitment to the rare disease communities we serve,” said Sarah Gonzalez, Head, Medical Diagnostics at Sanofi Genzyme, the specialty care global business unit of Sanofi. “While we have seen many significant advances in research over the past 30+ years, there are still tremendous challenges in helping patients get a diagnosis for many rare diseases.”
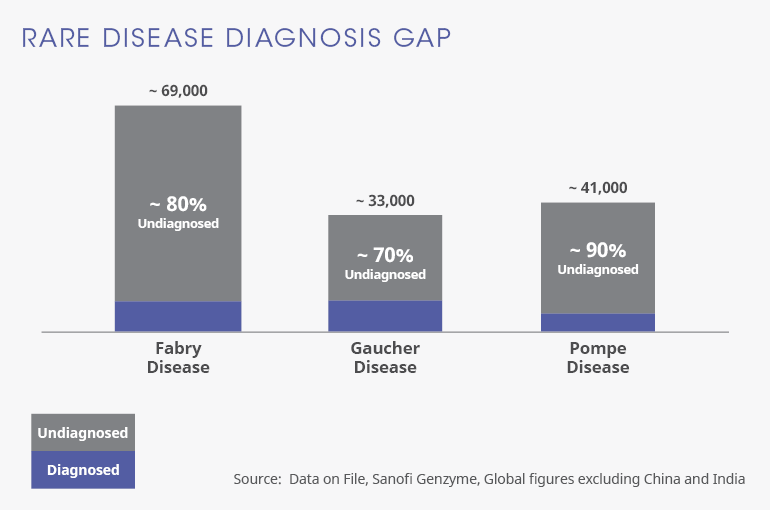
Barriers to Early Diagnosis
LSDs comprise a group of more than 40 genetic diseases. People living with LSDs experience a deficiency or malfunction in an enzyme needed to remove unnecessary waste material from cells in the body. These waste molecules accumulate in cells, disrupting cell function and leading to progressive organ damage.
LSDs are frequently characterized by a range of symptoms that can present at any time from early childhood to late in adulthood. Both disease progression and symptom severity can vary widely. The fact that these diseases are often rare and have symptoms associated with other more common diseases increases the risk of misdiagnosis, lengthy diagnostic delays and missed cases[1]. In addition to putting patients at risk of further disease progression, delay in diagnosis can lead to feelings of anxiety, frustration and isolation.
The Lantern Project
For more than a decade, Sanofi Genzyme has supported a range of programs to help patients have broader access to genetic testing services for specific types of LSDs.
The Lantern Project is specifically designed to overcome the barriers that patients experience in accessing comprehensive testing services for those diseases, including additional diagnostic tests that may be needed when results of genetic testing are suggestive but inconclusive.
One of the highlights of the program is the LGMD panel. This test offering is able to sequence 105 genes known to be associated with LGMDs and other myopathies, a heterogeneous group of muscular weakness disorders that vary in severity and age of onset. Using next generation sequencing technology that allows for rapid sequencing of multiple genes simultaneously, patients can be tested for multiple LGMD disorders at once in addition to other diseases that may cause similar symptoms including Pompe disease and spinal muscular atrophy.
Genetic testing can play an important role in confirming a diagnosis for patients living with many LSDs. Early diagnosis can also help patients advance to more timely initiation of options in disease management. The Lantern Project is not intended to and should not interfere in any way with a health care professional’s or patient’s independent judgment and freedom of choice in the treatment options for these diseases. Health care professionals and patients should always consider the full range of treatment options and select those most appropriate for the individual patient.
“The Lantern Project will help more patients access testing while also raising broader awareness of the critical need for more diagnostic services and support for all people affected by rare diseases,” said Gonzalez.
More information about The Lantern Project is available at LanternProjectDx.com.

Sources for Graphic
2 Mistry PK, Sadan S, Yang R, Yee J, Yang M. Consequences of diagnostic delays in type 1 Gaucher disease: the need for greater awareness among hematologists-oncologists and an opportunity for early diagnosis and intervention. Am. J. Hematol. 2007;82(8):697-701.
3 Data on File, Sanofi Genzyme market research/internal analysis
4 Germain DP et al, Orphanet Journal of Rare Diseases 2010, 5:30. Fabry Disease. 2010 22 Nov.
5 MPS Society. MPS I. 2017. Available at: http://mpssociety.org/mps/mps-I/. Accessed July 1, 2018.
6 Bruni S. et al. (2016) Mol. Geneti. Metab. The diagnostic journey of patients with mucopolysaccharidosis I: A real-world survey of patient and physician experiences.
[1] Epidemiology and diagnosis of lysosomal storage disorders; challenges of screening Kingma, Sandra D.K. et al. Best Practice & Research Clinical Endocrinology & Metabolism , Volume 29 , Issue 2 , 145 - 157