ORLANDO, Fla., June 25, 2018 /PRNewswire/ -- Sanofi (NYSE: SNY; EURONEXT: SAN) presented positive non-inferiority results of the BRIGHT study comparing its long-acting insulin Toujeo® to insulin degludec at the American Diabetes Association (ADA) 78th Scientific Sessions in Orlando, Florida.1
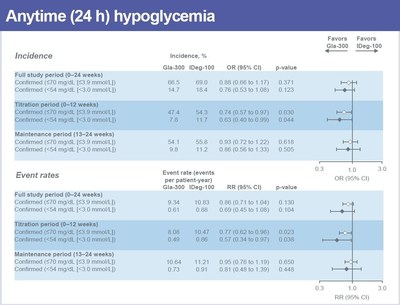
At the end of the BRIGHT study, Toujeo demonstrated comparable blood sugar (HbA1c) control versus insulin degludec (-1.64% vs. -1.59%, respectively). During the first 12 weeks of therapy, a period when patients and physicians work to determine the most appropriate individual insulin dose, Toujeo reduced the rate of low blood sugar (hypoglycemia) events by 23 percent and the incidence of low blood sugar events by 26 percent, compared to insulin degludec (p<0.05).2 During the subsequent 12 weeks of the study (treatment period: 13-24 weeks), the two treatments showed comparable incidence and rate of low blood sugar events.
"Hypoglycemia is a concern for people with diabetes, particularly in the initial period of dose adjustment," says Alice Cheng, Associate Professor of Endocrinology, University of Toronto, Toronto, Canada, and a primary investigator of the study. "Experiencing hypoglycemia, particularly in this early treatment period, could lead to patients discontinuing their treatment."
The incidence of low blood sugar at any time of the day during the 24-week treatment period was also comparable between Toujeo and insulin degludec (66.5% and 69.0%, respectively).
Results of the BRIGHT study:
The randomized controlled trial compared the efficacy and safety of Toujeo versus insulin degludec at 24 weeks. The study randomized 929 adults with type 2 diabetes to once-daily Toujeo or insulin degludec 100 Units/mL. Before randomization, participants were inadequately controlled with tablets (oral anti-hyperglycemic drugs, OADs), with or without a glucagon-like peptide-1 (GLP-1) receptor agonist, and had not previously started insulin therapy.1
The study met its primary endpoint demonstrating a reduction in blood sugar (HbA1c) levels comparable with Toujeo and insulin degludec from baseline to week 24 (at a non-inferiority margin of 0.3% and difference between treatments of -0.05% [95% CI −0.15 to 0.05%]).1
Over the 24-week period, incidence and event rates of confirmed (≤70 mg/dL and ≤54 mg/dL) low blood sugar (hypoglycemia) at any time of day were comparable between Toujeo and insulin degludec. During the titration period (0-12 weeks), confirmed low blood sugar event rates were lower by 23 percent (≤70 mg/dL) and 43 percent (≤54 mg/dL). In this period, the incidence of confirmed low blood sugar was also reduced by 26 percent (≤70 mg/dL) and 37 percent (≤54 mg/dL). Low blood sugar incidence and event rates were comparable in the maintenance time period (13-24 weeks).2
What is Toujeo® (insulin glargine injection) 300 Units/mL?
Prescription Toujeo® is a long-acting insulin used to control blood sugar in adults with diabetes mellitus.
- Toujeo® contains 3 times as much insulin in 1 mL as standard insulin (100 Units/mL)
- Toujeo® is not for use to treat diabetic ketoacidosis
- Toujeo® should not be used in children
Important Safety Information for Toujeo® (insulin glargine injection) 300 Units/mL
Do not take Toujeo® if you have low blood sugar or if you are allergic to insulin or any of the ingredients in Toujeo®.
Do not share your pen(s) with other people, even if the needle has been changed. You may give other people a serious infection, or get a serious infection from them.
Before starting Toujeo®, tell your doctor about all your medical conditions, including if you have liver or kidney problems, if you are pregnant or planning to become pregnant or if you are breastfeeding or planning to breastfeed.
Heart failure can occur if you are taking insulin together with pills called TZDs (thiazolidinediones), even if you have never had heart failure or other heart problems. If you have heart failure, it may get worse while you take TZDs with Toujeo®. Your treatment with TZDs and Toujeo® may need to be changed or stopped by your doctor if you have new or worsening heart failure. Tell your doctor if you have any new or worsening symptoms including:
- Shortness of breath
- Sudden weight gain
- Swelling of your ankles or feet
Tell your doctor about all the medications you take, including OTC medicines, vitamins, and supplements, and herbal supplements.
Toujeo® should be taken at the same time once a day. Test your blood sugar levels daily while using any insulin. Do not change your dose or type of insulin without talking to your doctor. Verify you have the correct insulin before each injection. Do NOT use a syringe to remove Toujeo® from your pen. Your dose for Toujeo® may be different from other insulins you have taken. Any change of insulin should be made cautiously and only under medical supervision.
Do NOT dilute or mix Toujeo® with any other insulin or solution. It will not work as intended and you may lose blood sugar control, which could be serious. Use Toujeo® only if the solution is clear and colorless with no particles visible.
While using Toujeo®, do not drive or operate heavy machinery until you know how Toujeo® affects you. Don't drink alcohol or use other medicines that contain alcohol.
The most common side effect of any insulin, including Toujeo®, is low blood sugar (hypoglycemia), which may be serious and life-threatening. Severe hypoglycemia may cause harm to your heart or brain. Symptoms of serious low blood sugar may include shaking, sweating, fast heartbeat, and blurred vision.
Toujeo® may cause severe allergic reactions that can lead to death. Get medical help right away if you have:
|
|
Toujeo® may have additional side effects including swelling, weight gain, low potassium, and injection site reactions which may include change in fat tissue, skin thickening, redness, swelling, and itching.
Toujeo® SoloStar® and Toujeo® Max SoloStar® are disposable prefilled insulin pens. It is important to perform a safety test when using a new pen for the first time. Talk to your doctor about proper injection technique and follow instructions in the Instruction Leaflet that comes with the pens.
Please see full Prescribing Information.
References
|
1. |
Cheng A, et al. Similar Glycemic Control and Less or Comparable Hypoglycemia with Insulin Glargine 300 U/mL (Gla-300) vs Degludec 100 U/mL (IDeg-100) in Insulin-Naïve T2DM on Antihyperglycemic Drugs ± GLP-1 RAs: The BRIGHT Randomized Study. Presentation 301-OR, American Diabetes Association (ADA) 78th annual congress in Orlando, Florida, U.S., June 25, 2018 |
|
2. |
Bolli GB, et al. Lower Hypoglycemia Rates with Insulin Glargine 300 U/mL (Gla-300) vs Insulin Degludec 100 U/mL (IDeg-100) in Insulin-Naïve Adults with T2DM on Oral Antihyperglycemic Therapy ± GLP-1 RA: The BRIGHT Randomized Study. Presentation 1032-P, American Diabetes Association (ADA) 78th annual congress in Orlando, Florida, U.S., June 23, 2018 |
About Sanofi
Sanofi is dedicated to supporting people through their health challenges. We are a global biopharmaceutical company focused on human health. We prevent illness with vaccines, provide innovative treatments to fight pain and ease suffering. We stand by the few who suffer from rare diseases and the millions with long-term chronic conditions.
With more than 100,000 people in 100 countries, Sanofi is transforming scientific innovation into healthcare solutions around the globe.
Sanofi, Empowering Life
|
US Media Relations |
Investor Relations Contact |
SOURCE Sanofi