PARIS, July 31, 2014 /PRNewswire/ -- Sanofi (NYSE: SNY; EURONEXT: SAN)
|
Q2 2014 |
Change (reported) |
Change (CER) |
|
|
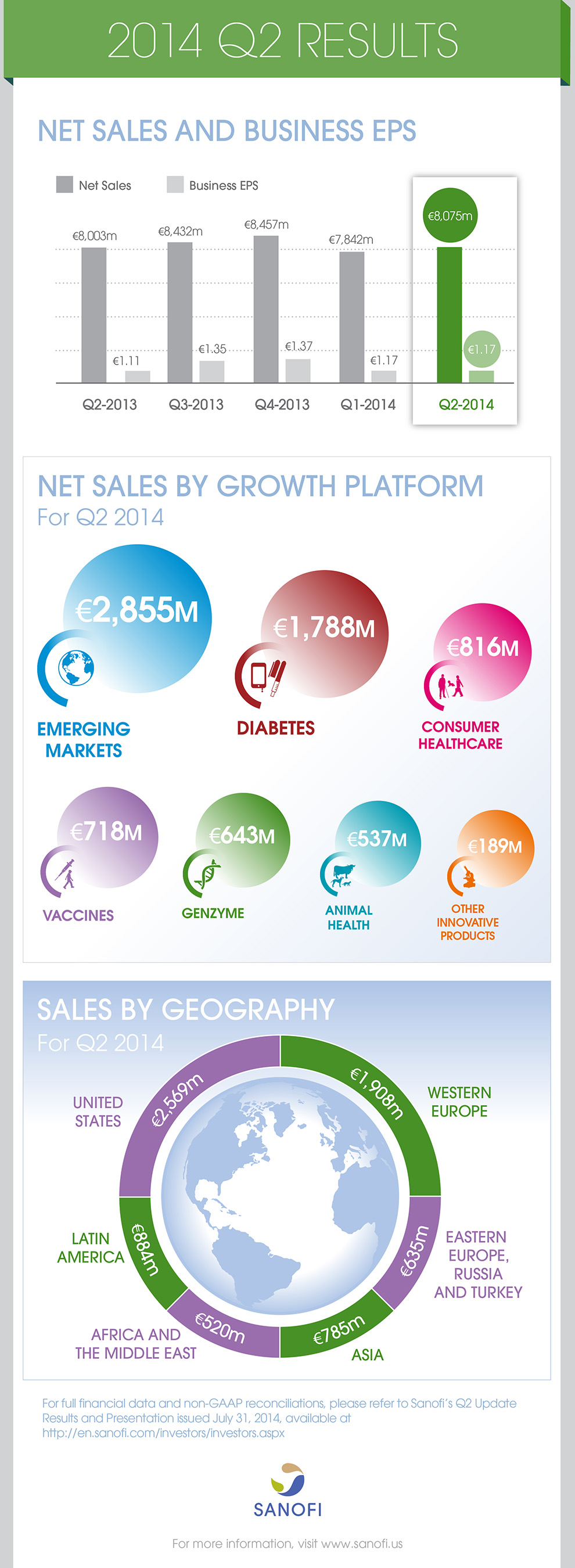
Net sales |
€8,075m |
+0.9% |
+6.4% |
|
Business net income(1) |
€1,537m |
+3.9% |
+13.0% |
|
Business EPS(2) |
€1.17 |
+4.5% |
+13.4% |
In order to facilitate an understanding of our operational performance, Sanofi comments on the business net income statement. Business net income(1) is a non-GAAP financial measure.(2) (EPS) Earnings Per Share
Commenting on the Group's performance in Q2 2014, Sanofi Chief Executive Officer, Christopher A. Viehbacher said, "Our solid second quarter performance reflects consistent execution of our growth strategy and allows us to slightly adjust upwards our 2014 financial guidance. This quarter, growth platforms represented more than 75% of our sales. Based on the solid momentum in our late stage pipeline, we are actively preparing for a wave of new product launches that will further redefine Sanofi as a biopharmaceutical leader."
To view the multimedia assets associated with this release, please click: http://www.multivu.com/mnr/7238451-sanofi-results-q2-2014
Q2 2014 Performance
- In the second quarter of 2014, Sanofi sales were €8,075 million, an increase of 0.9% on a reported basis. Exchange rate movements had a negative effect of 5.5 percentage points primarily reflecting the strength of the euro versus other currencies, in particular the U.S. Dollar.
- First-half sales reached €15,917 million, a decrease of 0.9% on a reported basis. Exchange rate movements had an unfavorable effect of 5.8 percentage points.
- Diabetes division sales grew 16.2% to €1,788 million in the second quarter driven by Lantus® (up 16.3% to €1,557 million). First-half sales of the Diabetes division grew 14.7% to €3,450 million.
- Second-quarter Consumer Healthcare products (CHC) recorded sales of €816 million, an increase of 20.2%. Several products (amounting €73 million in sales) previously recorded in prescription pharmaceuticals in the second quarter of 2013 were transferred to Consumer Healthcare products.
- Second-quarter sales of Genzyme grew 29.1% to €643 million, driven by the growth of Aubagio® and Fabrazyme®. Sales grew 27.3% (to €235 million) in the U.S., 47.3% (to €144 million) in Emerging Markets and 22.9% (to €209 million) in Western Europe. First-half sales of Genzyme increased 25.4% to €1,209 million.
- Other Innovative Products grew 13.3% to €189 million in the second quarter and 17.8% to €379 million in the first half, primarily driven by recent launches of Jevtana®, Zaltrap® and Auvi-Q™.
- Second-quarter consolidated sales of Sanofi Pasteur reached €718 million (down 0.4%) reflecting continued gradual recovery of Pentacel®, strong performance of Flu vaccines and Travel and Other Endemic vaccines which was offset by lower Booster vaccines sales. First-half consolidated sales of Sanofi Pasteur were €1,346 million, (down 2.2%).
- Second-quarter sales of Animal Health increased 6.2% to €537 million driven by the launch of NexGard™. In the U.S., Animal Health sales recorded strong growth (up 14.0% to €232 million). First-half sales of Animal Health increased 2.2% to €1,054 million.
R&D Update
- In July, the U.S. Food and Drug Administration (FDA) accepted for review the company's New Drug Application (NDA) for Toujeo®, an investigational basal insulin. This follows the acceptance of the marketing authorization dossier for Toujeo® by the European Medicines Agency (EMA) for EU countries in May 2014. In July, the NDA for Toujeo® was submitted to the Japanese Health Authorities (PMDA).
- In July, the Japanese Health Authority (PMDA) granted a marketing authorization for Jevtana® for the treatment of prostate cancer.
- In June, Genzyme submitted a new drug application in Japan for its oral Gaucher's disease treatment, Cerdelga™ (eliglustat tartrate).
- In May, Sanofi Pasteur's pediatric pentavalent vaccine Shan5™, developed and manufactured by its affiliate Shantha Biotechnics in Hyderabad, India, received prequalification status from the World Health Organization (WHO).
- In May, the U.S. Food and Drug Administration (FDA) accepted for review Genzyme's resubmission of its supplemental Biologics License Application (sBLA) seeking approval of Lemtrada™ (alemtuzumab) for the treatment of relapsing forms of multiple sclerosis. A six-month review period was assigned for the Lemtrada sBLA. Genzyme expects FDA action on the sBLA in the fourth quarter.
- At the end of July 2014, the R&D pipeline contained 46 projects (excluding Life Cycle Management) and vaccine candidates in clinical development of which 12 are in Phase III or have been submitted to the health authorities for approval.
To access the full press release of the Q2 2014 results, please click here: https://www.multivu.com/assets/7238451/documents/7238451-Q2-2014-results-original.pdf
Forward-Looking Statements
This press release contains forward-looking statements as defined in the Private Securities Litigation Reform Act of 1995, as amended. Forward-looking statements are statements that are not historical facts. These statements include projections and estimates and their underlying assumptions, statements regarding plans, objectives, intentions and expectations with respect to future financial results, events, operations, services, product development and potential, and statements regarding future performance. Forward-looking statements are generally identified by the words "expects", "anticipates", "believes", "intends", "estimates", "plans" and similar expressions. Although Sanofi's management believes that the expectations reflected in such forward-looking statements are reasonable, investors are cautioned that forward-looking information and statements are subject to various risks and uncertainties, many of which are difficult to predict and generally beyond the control of Sanofi, that could cause actual results and developments to differ materially from those expressed in, or implied or projected by, the forward-looking information and statements. These risks and uncertainties include among other things, the uncertainties inherent in research and development, future clinical data and analysis, including post marketing, decisions by regulatory authorities, such as the FDA or the EMA, regarding whether and when to approve any drug, device or biological application that may be filed for any such product candidates as well as their decisions regarding labelling and other matters that could affect the availability or commercial potential of such product candidates, the absence of guarantee that the product candidates if approved will be commercially successful, the future approval and commercial success of therapeutic alternatives, the Group's ability to benefit from external growth opportunities, trends in exchange rates and prevailing interest rates, the impact of cost containment policies and subsequent changes thereto, the average number of shares outstanding as well as those discussed or identified in the public filings with the SEC and the AMF made by Sanofi, including those listed under "Risk Factors" and "Cautionary Statement Regarding Forward-Looking Statements" in Sanofi's annual report on Form 20-F for the year ended December 31, 2013. Other than as required by applicable law, Sanofi does not undertake any obligation to update or revise any forward-looking information or statements.
About Sanofi
Sanofi, a global and diversified healthcare leader, discovers, develops and distributes therapeutic solutions focused on patients' needs. Sanofi has core strengths in the field of healthcare with seven growth platforms: diabetes solutions, human vaccines, innovative drugs, rare diseases, consumer healthcare, emerging markets, animal health and the new Genzyme. Sanofi is listed in Paris (EURONEXT: SAN) and in New York (NYSE: SNY).
Sanofi is the holding company of a consolidated group of subsidiaries and operates in the United States as Sanofi US. For more information on Sanofi US, please visit http://www.sanofi.us or call 1-800-981-2491.
Sanofi US is also on Twitter, Pinterest and Facebook. Visit us at https://twitter.com/SanofiUS, https://www.pinterest.com/sanofiUS and http://www.facebook.com/sanofiUS.
Media Relations:
908-989-0726
Email: USMediaRelations@Sanofi.com
Investor Relations:
908-981-5560
E-mail: IR@sanofi.com



To view the multimedia assets associated with this release, please click: http://www.multivu.com/mnr/7238451-sanofi-results-q2-2014
SOURCE Sanofi
